Analysis of Multi-Residue Pesticides in Vegetable Samples Using Solid-Phase Micro-Extraction (SPME) Coupled to Gas Chromatography-Electron-Capture Detector (GC-ECD)
Prashant Chaturvedi1, Rupender Kumari2, Rakesh Roshan Jha1 and Devendra Kumar Patel1*
Affiliation
1 Analytical Chemistry Laboratory, Regulatory Toxicology Group, Analytical Chemistry Laboratory & Regulatory Toxicology Group, CSIR Indian Institute of Toxicology Research, Vishvigyan Bhawan, 31, M. G. Marg, Lucknow, Uttar Pradesh, India
2 Toxicology Division, Central Forensic Science Laboratory, Kolkata, West Bengal, India
Corresponding Author
Dr. Devendra Kumar Patel, Analytical Chemistry Laboratory & Regulatory Toxicology Group, CSIR- Indian Institute of Toxicology Research, Vishvigyan Bhawan, 31, M. G. Marg, Lucknow - 226001, Uttar Pradesh, India, Tel: + 91-522-2627586-ext-228/ Fax: 91-522-2628227; E-mail: dkpatel@iitr.res.in
Citation
Patel, D.K., et al. Analysis of Multi-Residue Pesticides in Vegetable Samples Using Solid-Phase Micro-Extraction (SPME) Coupled to Gas Chromatography-Electron-Capture Detector (GC-ECD). (2018) J Analytic Bioanalytic Tech 3(1): 7- 13.
Copy rights
© 2018 Patel, D.K. This is an Open access article distributed under the terms of Creative Commons Attribution 4.0 International License.
Keywords
HS-SPME; Pesticides; Vegetable; GC-ECD
Abstract
A fast, simple, solvent less and inexpensive sample preparation method based on Head space solid-phase micro extraction (HS-SPME) coupled to gas chromatography-electron-capture detector (GC-ECD) is proposed for the determination of mix pesticide residues from vegetable samples. Polydimethylsiloxane (PDMS, 100 μm) fiber was used in this study. The mix pesticides (24 nos.) including organochlorines (15), organophosphates (06) and synthetic pyrethriods (03) were analysed in the vegetable samples (Cabbage, Tomato, Cauliflower, Chilli, Okra, Brinjal, Bottle gourd, Cucumber, Beetroot, Spinach, and Radish) collected from in and around of Lucknow city, India. Present study revealed the presence of HCH, DDT, methylparathion, malathion, chlorpyrifos, monocrotophos, endosulfan, cypermethrin, fenvalerate pesticides. Pesticides residue levels were compared with MRL fixed by Prevention of Food Adulteration Act (PFA), Govt. of India 1954. The method detection limits were in a linearity range of 0.003 - 0.024 mg/kg. A recovery percent varies from 86.1 to 96.4% with relative standard deviation (RSD) of 14%. The outcomes of the present study point towards the crucial need of implementing strict government’s rules related to food safety in order to put check on these kinds of health hazardous food contamination.
Introduction
Vegetables are important for the maintenance of health. They provide vitamins, antioxidants, enzymes, minerals and fiber. Daily consumption of at least 200 - 300 grams of fruits and 250 grams of vegetables is recommended for all age groups. Farmers relies on pesticides to protect crops from pests, weeds, rats, mice, flies and other insects from consuming and contaminating foods when they are being stored. India is the second largest user of pesticides in Asia. The use of pesticide in India is 0.33 kg/ hectare. This is against the pesticides consumption of 3.07 kg /hectare in France, 4.17 kg /hectare in Italy and 13.1 kg/ hectare in Japan and more than 15 kg/ hectare in United Kingdom, Canada and United States (Associated Chambers of Commerce and Industry of India (ASSOCHAM). To save 108 million tones of vegetables India applies 6000 tones of pesticides, about 15% of the total 40,000 tons consumed. According to Indian Pesticide report it is estimated that India approximately loses 18% of the crop yield valued at Rs.900 billion due to pest attack each year. Authors have reported the residues of OCs, OPs and SPs, along with fungicide and herbicides in fruit and vegetables from India[1-13].
Several methodologies have been developed for sample preparation prior to the analysis of pesticides from biological or environmental samples[14,15]. SPME was first developed in 1989 by Pawliszyn and coworkers and has been marketed by Supelco since 1993. Subsequently, the technique has grown enormously[16-18]. This technique integrates sampling, extraction, pre concentration and sample introduction in a simple single step procedure. Although SPME was initially introduced for the extraction of organic compounds from water samples[19], but, soon after, SPME have been successfully applied for the determination of pesticides in various complex sample matrices[20-28]. SPME is applied as described in several papers dealing with pesticide residue determination in fruits or in a large number of papers related to determination of volatile compounds in food commodities. SPME is the advanced micro extraction technique that is being increasingly used in the isolation and extraction of organic contaminants from environmental/food/biological samples. In this study, an easy and solvent less SPME method has been developed and validated followed by GC-ECD analysis for the simultaneous determination of multi-residue pesticides (24 Nos.) from raw vegetable samples.
Materials and Methods
In the present study 15 organochlorine pesticides (α-HCH, β-HCH,γ-HCH, δ-HCH, aldrin, diedrin, endrin, Alachlor, α-endosulfan, β-endosulfan, endosulfan sulfate, 4,4-DDE, 4,4-DDD, 2,4-DDT, 4,4-DDT) 06 organophosphate pesticides (Phorate , Monocrotophos, Metrhylparathion, Malathion Chlorpyrifos and Ethion) and 03 Pyrethyroids (cypermethrin, fenvalerate and deltamethrin) were analysed. The individual pesticide standards > 99% pure were procured from Sigma Aldrich (Bellefonte, PA, USA) and n-hexane (HPLC grade) was procured from Merck, India. Stock standard solutions of individual compounds (concentrations between 100 and 850 μg/ml) were prepared by exact weighing and dissolution in n-hexane. The mixed standard solution of 2 mg/ml concentration of each compound was prepared by dilution of the stock solutions with n-hexane. To avoid pesticide degradation the working standard solution was freshly prepared every week and stored at 4°C during the study. SPME holder, 1 cm long polydimethylsiloxane fiber (PDMS 100 μm), sampling stand, stirring hot plate, 4 ml amber glass vials (O.D. 15 mm x H45 mm) with PTFE/Silicone septa and a magnetic stirring flea were obtained from Sigma Aldrich (Bellefonte, PA, USA).
Instrumentation and experimental conditions
Perkin Elmer Clarus 500 Gas chromatograph equipped with Ni63 electron capture detector (GC-ECD) was used with a split less injector (0.75 mm I.D. glass liner, split less time) for thermal desorption. All the separations were carried out on Elite - 5 (30 m X 0.25 mm, 0.25 μm) capillary column (Perkin Elmer). The oven temperature was started at 200°C with 2min hold and then increased to 280°C at a rate of 2°C /min. The fiber depth in the injector was set to 2.5 cm and the thermal desorption time in the split less injector was 10 min at 280°C. The temperatures of the injector port and the detector were set at 280°C and 350°C, respectively. Ultrapure Nitrogen (99.999%) was used as the carrier gas with a flow rate of 0.79 ml /min.
Total 110 (10 samples of each variety) vegetable samples of Cabbage, Tomato, Cauliflower, Chilli, Okra, Brinjal, Bottle gourd, Cucumber, Beetroot, Spinach and Radish were collected from different market locations within Lucknow city, India (26° 55' N, 80° 59' E) in September-October 2011. Samples were procured and kept in refrigerator and analyzed next day of the collection.
10 gm of vegetable sample was homogenized with 10 ml of Milli-Q (Millipore, Milford, MA, USA) water using a high speed blender. Further 2ml of homogenate, 0.6 g of NaCl and magnetic flea were added in 4 ml amber glass vial capped with PTFE-faced silicon septa. PDMS (100 μm) fiber was then exposed in glass vial in headspace for 20 min at 80°C. Sample was stirred at 400 rpm constantly on a magnetic hotplate. After adsorption the syringe was removed from the vial and thermal desorption of the analytes was carried out by inserting the fiber into the GC injection port (at 280°C) for 10 minutes. Magnetic fleas were cleaned with detergent and water and finally rinsed with acetone for next set of SPME extraction.
Linear ranges and determination coefficients (r2) obtained for each pesticide, detection limit (LOD), limit of quantification (LOQ) are shown in Table 1.
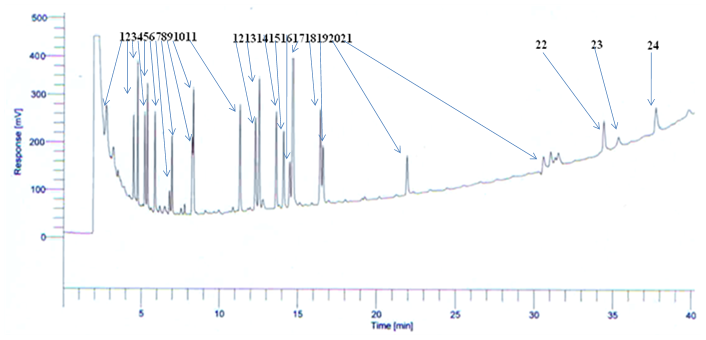
Recovery study was conducted by performing HS-SPME on the homogenate vegetable sample being spiked with mix pesticide standard solution at four-level concentrations as depicted in Table 1. Three replicate samples were used at each concentration. Control samples were processed along with spiked ones. GC-ECD chromatogram of HS-SPME recovered analytes from a homogenate vegetable sample being spiked mix pesticides standards solution at 0.02 mg/kg, is shown in (Figure 1). Recovery percentages corresponding to each targeted analytes were found in the range of 86.1 - 96.4, and linearity curve with R2 ranging between 0.9847 and 0.9986. (Table 1)
Figure 1: GC-ECD chromatogram of HS-SPME recovered analytes from a homogenate vegetable sample being spiked (0.002 mg/kg) with a mix standard solution of selected pesticides i.e. (1) Phorate, (2) α-HCH, (3) β-HCH, (4) γ-HCH, (5) δ-HCH (6) Methylparathion, (7) Malathion, (8) Monochrotophos, (9) Alachlor, (10) Chlorpyrifos, (11) Aldrin, (12) α-Endosulfan, (13) 4,4, DDE, (14) Dieldrin, (15) Endrin, (16) β-Endosulfan, (17) 4,4 DDD, (18) Ethion, (19) 2,4 DDT, (20) Endosulfan Sulfate, (21) 4,4 DDT, (22) Cypermethrin, (23) Fenvalerate, (24) Deltamethrin
Table 1: Performance evaluation of different pesticides in vegetable analyzed with HS-SPME-GC-ECD.
| Compound | Range (mg/kg) | R2 | RSD(%) | LOD (mg/kg) | LOQ (mg/kg) | Recovery (%) |
|---|---|---|---|---|---|---|
| &alphba;- HCH | 0.006-0.096 | 0.9890 | 11.5 | 0.002 | 0.006 | 94.2 |
| β- HCH | 0.006-0.096 | 0.9986 | 8.7 | 0.002 | 0.006 | 86.1 |
| γ- HCH | 0.006-0.096 | 0.9792 | 6.4 | 0.002 | 0.006 | 93.8 |
| δ- HCH | 0.006-0.096 | 0.9890 | 9.2 | 0.002 | 0.006 | 96.4 |
| Aldrin | 0.003-0.048 | 0.9892 | 8.4 | 0.001 | 0.003 | 88.9 |
| α-Endosulfan | 0.015-0.240 | 0.9987 | 10.8 | 0.005 | 0.015 | 90.1 |
| Dieldrin | 0.006-0.096 | 0.9992 | 8.5 | 0.002 | 0.006 | 94.5 |
| Alachlor | 0.006-0.096 | 0.9985 | 12.8 | 0.002 | 0.006 | 89.4 |
| 4,4’-DDE | 0.006-0.096 | 0.9943 | 9.7 | 0.002 | 0.006 | 88.9 |
| Endrin | 0.006-0.096 | 0.9890 | 10.1 | 0.002 | 0.006 | 91.7 |
| β -Endosulfan | 0.015-0.240 | 0.9797 | 11.9 | 0.005 | 0.015 | 88.2 |
| 4,4’-DDD | 0.006-0.096 | 0.9980 | 8.1 | 0.002 | 0.006 | 89.4 |
| 2-4’-DDT | 0.006-0.096 | 0.9981 | 7.8 | 0.002 | 0.006 | 87.1 |
| Endosulfan Sulfate | 0.015-0.240 | 0.9847 | 9.6 | 0.005 | 0.015 | 90.9 |
| 4,4’-DDT | 0.006-0.096 | 0.9915 | 10.6 | 0.002 | 0.006 | 93.7 |
| Phorate | 0.006-0.096 | 0.9948 | 9.7 | 0.002 | 0.006 | 94.9 |
| Methylparathion | 0.006-0.096 | 0.9962 | 9.4 | 0.002 | 0.006 | 89.1 |
| Malathion | 0.015-0.240 | 0.9986 | 8.5 | 0.005 | 0.015 | 91.8 |
| Chlorpyrifos | 0.003-0.048 | 0.9978 | 10.3 | 0.001 | 0.003 | 88.6 |
| Monocrotophos | 0.006-0.096 | 0.9991 | 13.8 | 0.002 | 0.006 | 87.2 |
| Ethion | 0.006-0.096 | 0.9928 | 11.4 | 0.002 | 0.006 | 93.7 |
| Cypermethrin | 0.015-0.240 | 0.9980 | 9.4 | 0.005 | 0.015 | 90.9 |
| Fenvalerate | 0.015-0.240 | 0.9983 | 8.6 | 0.005 | 0.015 | 89.4 |
| Deltamethrin | 0.015-0.240 | 0.9919 | 8.1 | 0.005 | 0.015 | 91.1 |
Optimizing conditions for extraction
Different parameters influencing HS-SPME were optimized such as extraction temperature, extraction time, sodium chloride amounts, desorption temperature, desorption time of the fiber in the GC injector port. All the conditions of SPME coupled to GC-ECD were systematically optimized using such an approach that the finally optimized conditions could support reasonable extraction of all the analytes of dissimilar volatilities.
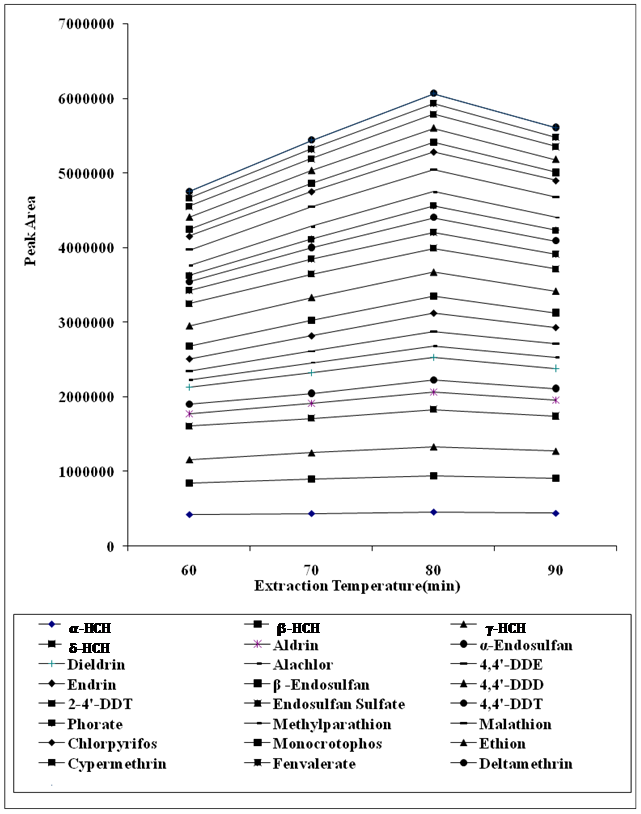
In order to study the temperature effect in spiked vegetable samples at 60, 70, 80 and 90°C and peak areas of target analytes were observed. Maximum area of analytes was achieved at 80°C because when the sample temperature is increased, the vapor pressure of the analyte and consequently the concentration of analyte in the headspace are increased. From a kinetic view, higher temperatures increase the transfer rate of the analytes from the matrix (e.g. water) to the headspace, while thermodynamically higher temperatures decrease the analytes partition coefficients between the headspace and fiber. Peak areas were decreased when temperature exceeded 80°C because adsorption is an exothermic process and disfavored at high temperature therefore 80°C was selected as working temperature for the analysis. (Figure 2)
Figure 2: Effect of extraction temperature on the peak areas of targeted analytes.
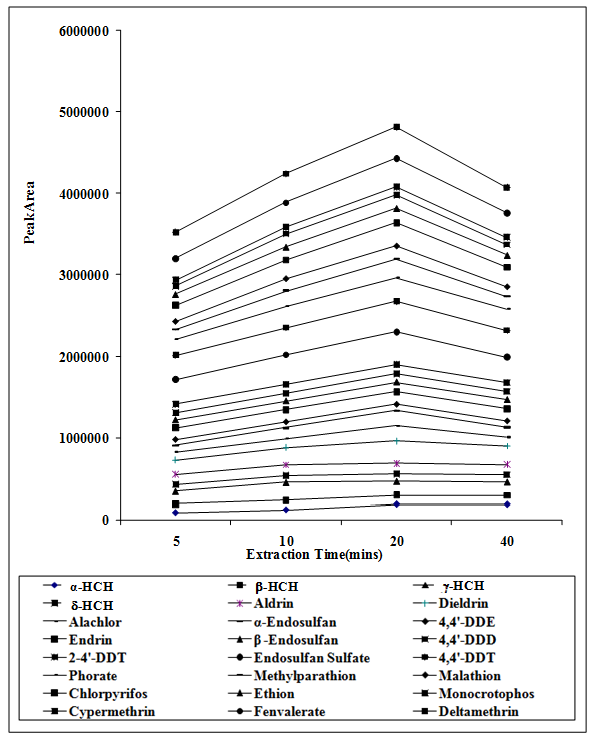
Extraction times were observed at different times between 05 - 40 mins. Maximum peak areas were observed when the extraction time was 20 mins. (Figure 3)
Figure 3: Effect of extraction time on the peak areas of targeted analytes.
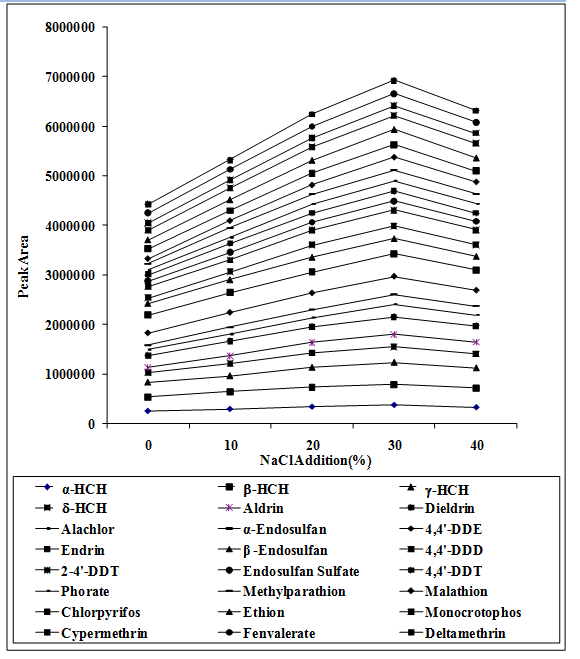
Spiked vegetable sample were treated with NaCl (w/v) 0, 10, 20, 30, 40%. The 30% (w/v) NaCl concentration was selected for extraction because peak areas of pesticides were declined when more than 30% NaCl (w/v) was added. Addition of sodium chloride reduced the solubility of target compounds in water by increasing higher distribution of the analytes in the headspace and onto the SPME fiber. The presence of higher concentrations of sodium chloride within the matrix may reduce the molecular movement of the target analytes, resulting in a decrease in extraction efficiency because excess concentration of NaCl deposited on SPME fiber. (Figure 4)
Figure 4: Effect of salt addition (NaCl) on the peak areas of targeted analytes.
Repeatability: The repeatability of the method performed under optimized conditions was determined by analyzing six samples of pesticides. The relative standard deviations of peak areas ranged from 5.4 – 8.5% and RSD < 14% observed for all compounds (Table 1)
Studying Recovery of Proposed Method: To evaluate extraction recoveries, successive extractions were performed on the single solution by the proposed fiber. After three successive extraction recoveries were found in the range of 86.1 – 96.4% (Table 1)
Results and Discussion
Residues of HCH, DDT, methylparathion, malathion, chlorpyrifos, monocrotophos, endosulfan, cypermethrin, fenvalerate pesticides were detected in vegetable samples (Table 2)
Table 2: Pesticide residues concentration in vegetable collected from different areas of Lucknow City.
| Vegetable | Samples Analysed | Pesticide | MRL (mg /kg) (PFA) | Mean Residue level (mg /kg) |
|---|---|---|---|---|
| Tomato | 10 | g-HCH | 1.0 | 0.37 |
| 10 | ΣDDT | 3.5 | 0.51 | |
| 10 | Malathion | 3.0 | 0.46 | |
| 10 | Chlorpyrifos | 0.2 | 0.28 | |
| 10 | Endosulfan | 2.0 | 0.49 | |
| 10 | Fenvalerate | 2.0 | 0.82 | |
| 10 | Cypermethrin | 0.2 | 0.12 | |
| Cabbage | 10 | ΣDDT | 3.5 | 0.50 |
| 10 | Methyl parathion | 1.0 | 0.79 | |
| 10 | Malathion | 3.0 | 0.62 | |
| 10 | Chlorpyrifos | 0.01 | 0.01 | |
| 10 | Endosulfan | 2.0 | 0.40 | |
| 10 | Cypermethrin | 2.0 | 0.28 | |
| 10 | Fenvalerate | 2.0 | 1.08 | |
| Cauliflower | 10 | ΣDDT | 3.5 | 0.38 |
| 10 | Methyl parathion | 1.0 | 0.85 | |
| 10 | Malathion | 3.0 | 0.88 | |
| 10 | Chlorpyrifos | 0.01 | 0.01 | |
| 10 | Endosulfan | 2.0 | 0.32 | |
| 10 | Fenvalerate | 2.0 | 0.55 | |
| Chilli | 10 | g-HCH | 1.0 | 0.22 |
| 10 | Methyl parathion | 1.0 | 0.55 | |
| 10 | Malathion | 3.0 | 0.57 | |
| 10 | Chlorpyrifos | 0.2 | 0.12 | |
| 10 | Endosulfan | 2.0 | 0.51 | |
| Okra | 10 | ΣDDT | 3.5 | 0.35 |
| 10 | Methyl parathion | 1.0 | 0.21 | |
| 10 | Malathion | 3.0 | 0.52 | |
| 10 | Chlorpyrifos | 0.2 | 0.36 | |
| 10 | Endosulfan | 2.0 | 0.29 | |
| 10 | Cypermethrin | 0.2 | 0.08 | |
| Brinjal | 10 | g-HCH | 1.0 | 0.11 |
| 10 | Malathion | 3.0 | 0.41 | |
| 10 | Endosulfan | 2.0 | 0.38 | |
| 10 | Cypermethrin | 0.2 | 0.26 | |
| 10 | Fenvalerate | 2.0 | 0.31 | |
| Bottle gourd | 10 | ΣDDT | 3.5 | 0.37 |
| 10 | Methyl parathion | 1.0 | 0.72 | |
| 10 | Malathion | 3.0 | 2.16 | |
| 10 | Chlorpyrifos | 0.2 | 0.27 | |
| 10 | Endosulfan | 2.0 | 0.34 | |
| 10 | Fenvalerate | 2.0 | 0.91 | |
| Cucumber | 10 | Methyl parathion | 1.0 | 0.64 |
| 10 | Malathion | 3.0 | 0.98 | |
| 10 | Chlorpyrifos | 0.2 | 0.21 | |
| 10 | Endosulfan | 2.0 | 0.42 | |
| 10 | Cypermethrin | 0.2 | 0.16 | |
| 10 | Fenvalerate | 0.2 | 0.14 | |
| Beetroot | 10 | Methyl parathion | 1.0 | 0.75 |
| 10 | Malathion | 3.0 | 2.18 | |
| 10 | Chlorpyrifos | 0.2 | 0.15 | |
| 10 | Endosulfan | 2.0 | 0.57 | |
| 10 | Cypermethrin | 0.2 | 0.15 | |
| Spinach | 10 | Malathion | 3.0 | 2.80 |
| 10 | Endosulfan | 2.0 | 0.46 | |
| 10 | Cypermethrin | 0.2 | 0.17 | |
| 10 | Fenvalerate | 2.0 | 0.05 | |
| Radish | 10 | Malathion | 3.0 | 2.18 |
| 10 | Chlorpyrifos | 0.2 | 0.28 | |
| 10 | Endosulfan | 2.0 | 0.53 | |
| 10 | Cypermethrin | 0.2 | 0.24 | |
| 10 | Fenvalerate | 2.0 | 0.19 |
γ-HCH was detected in 28 out of the 110 samples. The average lower concentration was 0.11 mg/kg in Brinjal samples while higher mean concentration was 0.37 mg/kg found in Tomato samples. The residues of γ-HCH were above MRL in 09 samples as per PFA Act.
ΣDDT was detected in 21 out of the 110 samples. The average lower concentration was 0.35 mg/kg in Okra samples while average higher concentration was 0.51 mg/kg found in Tomato samples. The residues of γ-HCH were above MRL in 07 samples as per PFA Act.
Methyparathion was detected in 38 out of the 110 samples. The average lower concentration was 0.21 mg/kg in Okra samples while higher concentration was 0.85 mg/kg found in Cauliflower samples. The residues of methyparathion were above MRL in 12 samples.
Malathion residues were found in 47 samples out of the 110. Minimum average concentration was found 0.41 in case of Brinjal samples and maximum concentration 0.62 mg/kg was found in cabbage samples. The residues of Malathion were below maximum residue level.
Chlorpyrifos residues were detected in 49 out of the 110 samples. The average lower concentration was 0.01 mg/kg in cabbage samples and high 0.153 mg/kg was found in okra samples. The residues of chlorpyrifos were above MRL in 08 samples.
Total Endosulfan (α-Endosulfan, β-Endosulfan and Endosulfan sulfate) was found in 64 vegetable samples. Lowest average concentration was 0.29 mg/kg in Okra samples while highest concentration was 0.57 mg/kg in Beetroot samples. The residues of Endosulfan were above MRL in 17 samples.
Cypermethrin was detected in 48 out of the 110 samples. The average lower concentration was 0.08 mg/kg in Okra samples while higher concentration was 0.28 mg/kg found in Cabbage samples. The residues of Cypermethrin were above MRL in 12 samples.
Fenvalerate residues were detected in 39 out of the 110 samples. The average lower concentration was 0.31 mg/kg in Brinjal samples and high 1.08 mg/kg was found in Cabbage samples. The residues of Fenvalerate were above MRL in 19 samples as per PFA Act.
Conclusion
A periodical monitoring of pesticide residues in vegetables are the recent need for the consumers. Restricting the spray of pesticides can reduce the contamination level. A fast, simple, solvent free Headspace SPME-GC method can be used instead of the conventional extraction technique for the determination and identification of organochlorine, organophosphate and pyrethroid pesticides in vegetable samples.
Acknowledgement: The authors wish to thank the Director, Council of Scientific and Industrial Research – Indian Institute of Toxicology Research (CSIR-IITR), Lucknow, India, for providing the necessary facilities for this research study.
Competing interest: The authors do not have any relevant affiliations, financial involvement and conflicts with any organization and person with the subject matter and material discussed in the manuscript. The authors were not influenced by, or may be perceived to be influenced by, any personal or financial relationship with other people or organizations for the interpretation of data or presentation of information depicted in the present study. The author did not get any writing assistance for the production of this manuscript.
Abbreviations: HCH: Hexachlorocyclohexane, α: Alpha, β: Beta, γ: Gamma, δ: Delta, DDE: Dichloro Diphenyl dichloroethylene, DDD: Dichloro Diphenyl dichloroethane, DDT: Dichloro Diphenyl trichloroethane, PDMS: Polydimethylsiloxane, OCs: Organochlorines, Ops: Organophosphorus, HS – SPME: Headspace Solid-Phase Micro-Extraction, RPM: Rounds per minute, GC - ECD - Electron-Capture Detector, Ni63- Nickel63
References
- 1. Arthur, C.L., Pawliszyn, J. Solid phase microextraction with thermal desorption using fused silica optical fibers. (1990) Anal Chem 62(19): 2145-2148.
- 2. Aulakh, J.S., Malik, A.K., Kaur, V., et al. A review on solid phase micro extraction high performance liquid chromatography (SPMEHPLC) analysis of pesticides. (2005) Crit Rev Anal Chem 35: 71–85.
- 3. Barcelo, D., Hennion, M.-C. On-line sample handling strategies for the trace-level determination of pesticides and their degradation products in environmental waters. (1995) Anal Chim Acta 318(1): 1-41.
PubMed||CrossRef||Others
- 4. Belardi, R.P. Pawliszyn, J. Application of chemically modified fused silica fibers in the extraction of organics from water matrix samples and their rapid transfer to capillary columns. (1989) Water Qaulity Res J Can 24(1): 179-191.
PubMed||CrossRef||Others
- 5. Beltran, J., Lopez, F.J., Hernandez, F. Solid-phase micro-extraction in pesticide residue analysis. (2000) J Chromatogr A 885(1-2): 389–404.
- 6. Beltran, J., Peruga, A., Pitarch, E., et al. Application of solid-phase microextraction for the determination of pyrethroid residues in vegetable samples by GC-MS. (2003) Anal Bioanal Chem 376(4): 502-511.
- 7. Bhanti, M., Taneja, A. Monitoring of organochlorine pesticide residues in summer and winter vegetables from Agra, India-a case study. (2005) Environ Monit Assess 110(1-3): 341–346.
- 8. Bianchi, F., Careri, M., Musci, M. Fish and food safety: Determination of formaldehyde in 12 fish species by SPME extraction and GC–MS analysis. (2007) Food Chem 100(3): 1049-1053.
- 9. Dikshit, T.S.S., Raizada, R.B., Kumar, S.N., et al. Residues of DDT and HCH in major source of drinking water in Bhopal, India. (1990) Bull Environ Contam Toxicol 45(3): 389–393.
- 10. Frank, R., Braun, H.E., Ripley, B.D. Residue of insecticide, fungicides and herbicide in fruits produced in Ontario, Canada. (1987) Bull Environ Contam Toxicol 39(2): 272–279.
- 11. Holt, R. Mechanisms Effecting analysis of volatile flavour components by solid-phase microextraction and gas chromatography. (2001) J Chromatogr A 937(1-2): 107-114.
- 12. Kataoka, H., Lord, H.L., Pawliszyn, J. Applications of solid-phase microextraction in food analysis. (2000) J Chromatogr A 880(1-2): 35–62.
- 13. Kumari, B., Gulati, R., Kathpal, T.S. Monitoring of pesticidal contamination in honey. (2003a) The Korean J Apicult 18(2): 155–160.
PubMed||CrossRef||Others
- 14. Kumari, B., Kathpal, T.S. Monitoring of pesticide residues in vegetarian diet. (2008) Environ Monit Assess 151(1-4): 19-26.
- 15. Kumari, B., Kumar, R., Madan, V.K., et al. Magnitude of pesticidal contamination in winter vegetables from Hisar, Haryana. (2003b) Environ Monit Assess 87(3): 311–318.
- 16. Kumari, B., Madan, V.K., Kathpal, T.S. Monitoring of pesticide residues in fruits. (2006) Environ Monitor Assess 123(1-3): 407–441.
PubMed||CrossRef||Others
- 17. Kumari, B., Madan, V.K., Kumar, R., et al. Monitoring of seasonal vegetables for pesticide residues. (2002) Environ Monit Assess 74(3): 263–270.
PubMed||CrossRef||Others
- 18. Kumari, B., Madan, V.K., Singh, J., et al. Monitoring of pesticidal contamination of farmgate vegetables from Hisar. (2004) Environ Monit Assess 90(1-3): 65–77.
- 19. Kumari, B., Singh, J., Singh, S., et al. Monitoring of butter and ghee (clarified butter fat) for pesticidal contamination from cotton belt of Haryana, India. (2005) Environ Monitor Assess 105(1-3): 111–120.
- 20. Lamprodon, D.A., Albanis, T.A. Headspace solid-phase microextraction applied to the analysis of organophosphorus insecticides in strawberry and cherry juices. (2002) J Agric Food Chem 50(12): 3359-3365.
- 21. Matich, A.J., Rowan, D.D., Banks, N.H. Solid-phase microextraction for quantitative headspace sampling of apple volatiles. (1996) Anal Chem 68(23): 4114-4118.
- 22. Mestres, M., Busto, O. Headspace solid-phase microextraction analysis of volative sulphides and disulphides in wine aroma. (1998) J Chromatogr A 808(1-2): 211-218.
- 23. Pagliuca, G., Gazzotti, T., Zironi, E., et al. Residue analysis of organophosphorus pesticides in animal matrices by dual column capillary gas chromatography with nitrogen–phosphorus detection. (2005) J Chromatogr A 1071(1-2): 67-70.
- 24. Quintero, A., Caseiies, M.J., Ettiene, G., et al. Monitoring of organphosphorus pesticide residues in vegetables of agricultural area in Venezuela. (2008) Bull Environ Contam Toxicol 81(4): 393–396.
- 25. Shahi, D.K., Nisha, K., Sharma, A. Monitoring of pesticide residues in market vegetable at Ranchi, Jharkhand (India). (2005) J Environ Sci Eng 47(4): 322–325.
PubMed||CrossRef||Others
- 26. Song, J., Gardner, B.D., Holland, J.F., et al. Rapid analysis of volatile flavour compounds in apple fruit using SPME and GC/Time-of-Flight Mass Spectrometry. (1997) J Agricult Food Chem 45: 1801-1807.
- 27. Song, J., Fan, L., Beaudry, R.M. Application of solid-phase microextraction and Gas Chromatography/Time-of-Flight Mass Spectrometry for rapid analysis of Flavour volatiles in tomato and strawberry fruits. (1998) J Agricult Food Chem 46: 3721-3726.
- 28. Wang, L., Yongchao, L., Xin, J. Analysis of eight organphosphorus pesticide residues in fresh vegetables retailed in agricultural product market of Nanjing, China. (2008) Bull Environ Contam Toxicol 81(4): 377–382.